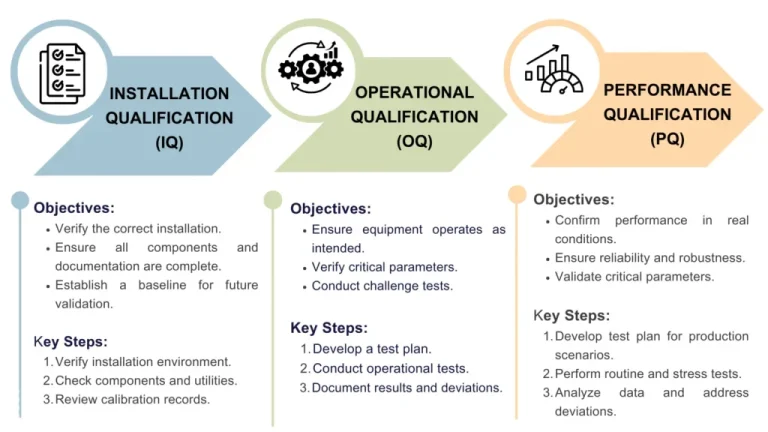
Full validation review
EvoSafe conducts Full Validation Reviews to assess the completeness, accuracy, and compliance of manufacturing validation activities. We review validation plans, protocols, execution results, and documentation to confirm alignment with GxP, FDA, and industry standards.
Our review identifies gaps, risks, and improvement opportunities—helping organizations strengthen compliance, maintain a validated state, and prepare confidently for audits and inspections.

Services
- Review of IQ/OQ/PQ protocols and reports
- Audit and inspection readiness
- Validation documentation and records review
- Regulated manufacturing environments overview
- GMP - Good Manufacturing Processes review
